Is Peptide Therapy FDA-Approved? The Honest Truth
The answer to 'is peptide therapy FDA-approved' isn't a simple yes or no. Learn the key differences between approved drugs and research peptides.

This content is for informational purposes only and does not constitute medical advice.
The question is peptide therapy FDA-approved is one of the most common we hear, and the answer isn't a simple yes or no. The reality is complex. Some specific peptide drugs, like Ozempic for weight loss, have gone through rigorous testing and are fully FDA-approved for certain conditions. However, many other peptides used for healing, anti-aging, and performance are not individually FDA-approved. They are often prepared by special pharmacies, a process called compounding. Understanding this difference is crucial for making safe and informed decisions about your health in 2026.
General Questions About FDA Approval & Peptide Therapy
What is the simple answer to "is peptide therapy FDA-approved"?
The answer is mixed. Specific peptide medications are FDA-approved, but the broad practice of "peptide therapy" is not. Think of it like this: the FDA approves individual drugs, not entire categories of treatment. For example, semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) are FDA-approved peptide drugs for diabetes and weight management. However, peptides like BPC-157, often used for tissue repair, have not gone through the same approval process. Many of these are accessed through compounding pharmacies under a doctor's prescription, which operates under a different set of regulations (PeptideClinicLocator.com).
How many peptides are FDA-approved in 2026?
As of early 2026, there are 49 FDA-approved peptide active ingredients on the market (Pure Peptide Clinic). These approved peptides are formulated into well-known drugs that have undergone extensive clinical trials to prove their safety and effectiveness for specific medical conditions. This list includes long-standing medications like insulin for diabetes and newer drugs that have seen a massive surge in popularity. The global market for these approved peptide drugs was valued at over $52 billion in 2025, showing just how significant this class of medication has become in modern medicine.
What does "FDA-approved" actually mean for a peptide?
FDA approval means a specific peptide drug has passed a long and expensive review process to prove it is safe and effective for its intended use. This process, known as a New Drug Application (NDA) or Biologics License Application (BLA), requires multiple phases of clinical trials, often involving thousands of patients (PeptideClinicLocator.com). For example, clinical trials for GLP-1 agonists like semaglutide involved over 15,000 participants (Peptide Publicus). This high bar ensures the drug's benefits outweigh its risks for a specific condition and dosage. Only large pharmaceutical companies can typically afford this process.
What is a compounded peptide?
A compounded peptide is a medication prepared by a licensed compounding pharmacy for an individual patient. A doctor writes a specific prescription, and the pharmacy creates the medication, often by combining or altering ingredients to meet the patient's unique needs. This is different from mass-produced drugs from a large manufacturer. Compounding is legal and regulated, but the compounded medications themselves are not individually FDA-approved. They are made using bulk drug substances that appear on an FDA-approved list, ensuring a baseline level of quality and safety for the core ingredients.
Are compounded peptides FDA-approved?
No, compounded peptides are not FDA-approved. While the active pharmaceutical ingredients used by licensed compounding pharmacies must meet certain standards, the final mixed product has not undergone the rigorous, large-scale clinical trials required for formal FDA approval. This is the main distinction. FDA approval applies to a specific, branded drug product, not the customized preparations made by a compounding pharmacy for an individual patient based on a doctor's prescription. Their safety is overseen by state boards of pharmacy and relies on the quality of the pharmacy itself.
Why are some peptides FDA-approved and others aren't?
The main reasons are time and money. The FDA's New Drug Application process is extremely expensive, often costing hundreds of millions or even billions of dollars, and it can take over a decade to complete. This is usually only feasible for large pharmaceutical companies that can patent a new molecule and expect a significant financial return, like the over $20 billion annual market for drugs like semaglutide (Peptide Publicus). Many other promising peptides either lack this massive profit potential or haven't been studied extensively enough in large-scale human trials to warrant that investment, so they remain in the compounded or research space.
What changed with peptide regulations in 2026?
In February 2026, Health and Human Services (HHS) Secretary Robert F. Kennedy Jr. announced a significant policy shift. The plan is to reclassify approximately 14 peptides, including popular ones like BPC-157 and Ipamorelin, from Category 2 (banned for compounding) back to Category 1 (allowed for compounding). This move aims to reverse a 2023 FDA decision that had restricted access due to safety concerns like immunogenicity risk (Peptide-db.com). The goal is to provide a safer, regulated supply through licensed pharmacies instead of pushing consumers toward the risky, unregulated "research chemical" market that emerged after the ban.
Are the newly reclassified peptides now FDA-approved?
No, they are not. It's a critical distinction. The reclassification, once officially published by the FDA, simply allows licensed compounding pharmacies to legally prepare these peptides for patients again. It does not mean these peptides, such as BPC-157, have received FDA approval. This move restores a regulated supply chain with quality controls, but it does not change the fact that these specific peptides have not completed the extensive human clinical trials needed to be sold as FDA-approved drugs (Peptide-db.com). Patients still need a prescription and should work with a knowledgeable doctor.
Not sure which treatment is right for you, or how to navigate these regulations? A qualified provider can help. Take our free quiz to get matched with a provider who understands your goals.
Why would someone choose a non-FDA-approved compounded peptide?
People often choose compounded peptides for three main reasons: access, customization, and cost. Many peptides showing promise for wellness, anti-aging, or tissue repair are not FDA-approved, so compounding is the only legitimate medical channel to access them. Doctors can also prescribe customized doses or combinations not available in commercial drugs. Finally, cost is a huge factor. An FDA-approved drug like semaglutide can cost over $1,000 a month, while a compounded peptide like CJC-1295 with Ipamorelin might cost $100–$300 per month (Peptide Publicus).
What's the difference between compounded and "research only" peptides?
The difference is massive and comes down to safety, legality, and quality. Compounded peptides are prepared by a licensed and regulated U.S. pharmacy for a specific patient with a doctor's prescription. "Research Use Only" peptides are sold online, often illegally, without a prescription and are not intended for human consumption. These products have no guarantee of purity, dosage, or safety and could be contaminated. The FDA actively issues warning letters to companies marketing these as drugs, as seen in the December 2024 letter to Prime Peptides (FDA.gov).

Is Peptide Therapy FDA-Approved for Specific Conditions?
Is peptide therapy FDA-approved for weight loss?
Yes, specific peptide drugs are FDA-approved for weight loss. The most famous examples are the GLP-1 receptor agonists. Semaglutide is the active ingredient in Ozempic (approved for type 2 diabetes) and Wegovy (approved for weight management). Similarly, tirzepatide is the active ingredient in Mounjaro (for diabetes) and Zepbound (for weight management). These drugs have proven highly effective in large clinical trials (Peptide Publicus). While these specific drugs are approved, not all peptides used for weight loss have gone through the same process.
Is peptide therapy FDA-approved for anti-aging?
Generally, no. The FDA does not approve drugs specifically for "anti-aging," as it is not considered a medical condition. However, some FDA-approved peptides are used "off-label" for goals related to aging. For example, Tesamorelin (brand name Egrifta) is FDA-approved to reduce excess visceral fat in HIV patients with lipodystrophy (CalcMyPeptide). Because it stimulates growth hormone, some doctors prescribe it off-label for age-related conditions. Other peptides popular for aging, like those explored in our guide to aging supplements, are typically available only through compounding pharmacies.
Is peptide therapy FDA-approved for healing and recovery?
No. Currently, there are no FDA-approved peptides specifically for general tissue repair or accelerated injury healing in the general population. Peptides like BPC-157 and TB-500 are popular for these uses but have not undergone the required clinical trials for FDA approval. Their safety and efficacy data come mostly from animal studies and anecdotal reports from clinicians (Peptide Publicus). Once the pending regulatory changes are finalized, peptides like BPC-157 are expected to be available again through compounding pharmacies, providing a safer alternative to the unregulated online market.
Is peptide therapy FDA-approved for skin and beauty?
Yes, a new approval in 2026 marks a big step forward. In March 2026, the FDA approved ICOTYDE (icotrokinra), a first-of-its-kind oral peptide for moderate-to-severe plaque psoriasis (MarketMinute). This is a major development for dermatological conditions. Other peptides used for cosmetic purposes, like GHK-Cu, are popular for their potential skin-tightening and regenerative properties but are not FDA-approved drugs. They are often found in topical serums or can be prescribed as an injectable through a compounding pharmacy. For more on this, see our guide to the best injectable peptides for skin tightening.
Is peptide therapy FDA-approved for sexual health?
Yes, at least one peptide is FDA-approved for a specific aspect of sexual health. Bremelanotide, sold under the brand name Vyleesi, is an FDA-approved injectable peptide used to treat hypoactive sexual desire disorder (HSDD) in premenopausal women. It is one of only a handful of approved drugs for this condition. Other peptides, such as PT-141 (a variant of bremelanotide), are often used for erectile dysfunction and libido enhancement but are typically sourced through compounding pharmacies and are not individually FDA-approved.
Compounded Peptides & Safety: Is This Type of Peptide Therapy FDA-Approved?
How are compounded peptides regulated?
Compounded peptides are not directly regulated by the FDA in the same way as commercial drugs. Instead, they fall under the authority of state boards of pharmacy, which set standards for quality, safety, and operational procedures. The FDA sets rules about which bulk drug substances can be used in compounding. In 2023, the FDA restricted many peptides by placing them on a "Category 2" list, making them ineligible for compounding. The expected 2026 reclassification would move many of these back to "Category 1," allowing licensed pharmacies to legally prepare them again with a valid prescription (PeptideMark).
Are compounded peptides safe?
The safety of compounded peptides depends heavily on the quality of the compounding pharmacy. Reputable, licensed pharmacies follow strict sterile compounding procedures and use high-quality, tested ingredients. This is why the potential 2026 reclassification is so important, as it would shift users from risky online "research" sites to a regulated medical supply chain (ProPublica). However, because these peptides lack large-scale human safety data, working with an experienced physician is essential. They can monitor your progress and help manage any potential side effects. You can learn more about finding a legitimate peptide clinic here.
Want to see which peptide options fit your budget and personal health goals? Our free quiz takes 3 minutes and matches you with vetted providers in your state.
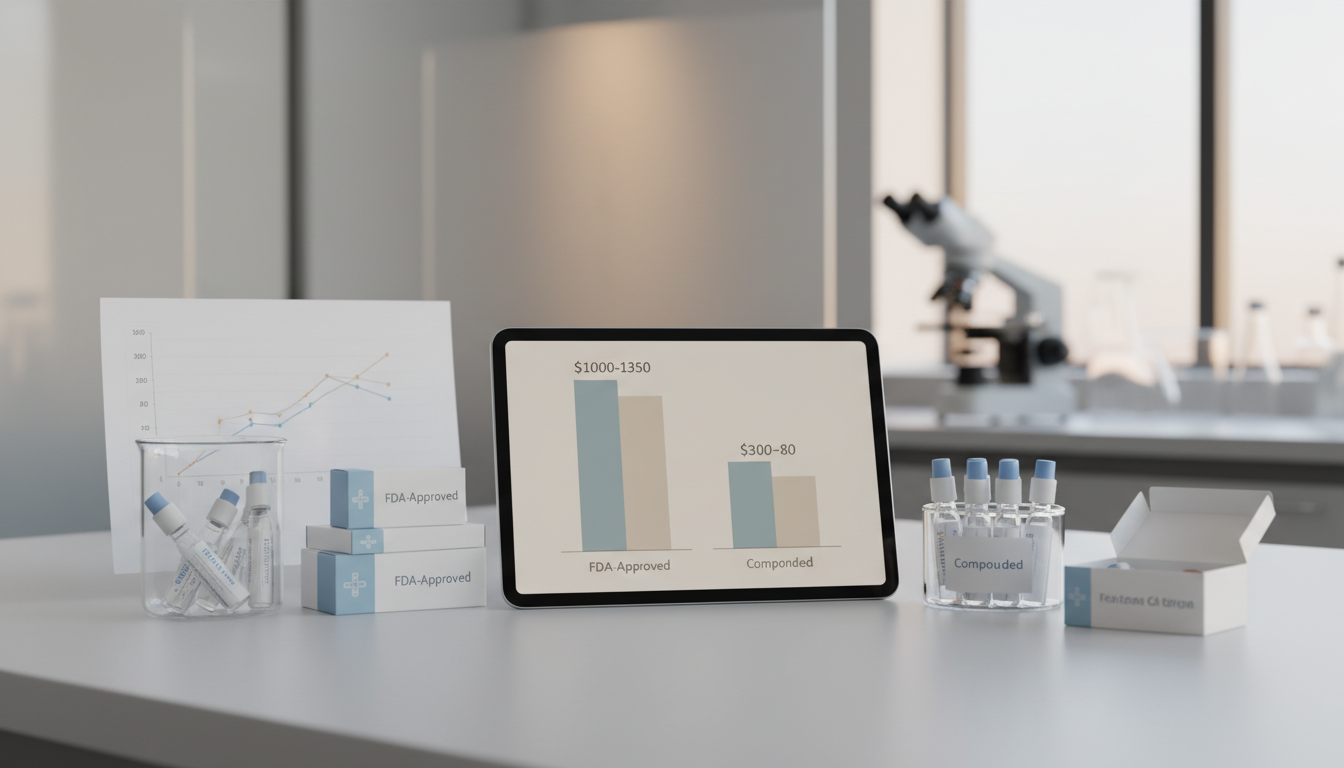
How much do compounded peptides cost compared to FDA-approved ones?
The cost difference is significant. FDA-approved peptide drugs for weight loss, like Wegovy or Zepbound, can cost between $1,000 and $1,350 per month without insurance coverage (Peptide Publicus). In contrast, popular compounded peptides for wellness, healing, or anti-aging typically range from $100 to $300 per month for the peptide itself. When you factor in consultations and supplies, the total monthly cost for a compounded peptide protocol often lands between $300 and $800. This lower price point makes them much more accessible for many people seeking these therapies. For a deeper look, check out our guide on peptide therapy costs.
How do I find a safe and legitimate source for peptides?
The only safe and legal way to access therapeutic peptides is through a licensed medical provider who writes a prescription to a reputable U.S.-based compounding pharmacy. Never buy products labeled "for research use only" for personal use. To find a reliable provider, look for clinics with experienced medical staff who specialize in hormone and peptide therapy. They should conduct a thorough health evaluation, including blood work, before prescribing any treatment. You can use online directories or platforms that vet peptide clinics to ensure you are connecting with qualified professionals.
The Bottom Line
So, is peptide therapy FDA-approved? The answer remains nuanced in 2026. While a growing number of powerful peptide drugs have earned FDA approval for specific diseases, many others used for general wellness and recovery exist in a separate, regulated space through compounding pharmacies. The anticipated regulatory shift to allow compounding of peptides like BPC-157 and Ipamorelin is a positive step toward safety and accessibility, but it is not the same as FDA approval. Always work with a qualified healthcare provider to discuss the risks and benefits.
Ready to explore your options with a professional? Take our free 3-minute quiz to get matched with a licensed provider who can design a plan based on your unique health goals.
FAQs
What peptides were reclassified in 2026?
As of April 2026, the formal FDA list has not yet been published. However, the HHS announcement in February 2026 indicated approximately 14 peptides are expected to move from Category 2 to Category 1, allowing them to be legally compounded. The most anticipated names on this list include BPC-157, CJC-1295, Ipamorelin, AOD-9604, Thymosin Alpha-1, Selank, and Semax (Peptide-db.com).
Is BPC-157 FDA-approved?
No, BPC-157 is not an FDA-approved drug. It has not gone through the large-scale human clinical trials required for approval. However, due to its popularity for tissue repair and recovery, it is one of the key peptides expected to be reclassified in 2026, which would once again allow licensed compounding pharmacies to prepare it with a doctor's prescription.
Is it legal to buy peptides online?
It is illegal for companies to market and sell peptides for human consumption without FDA approval or a prescription. Peptides sold online are often labeled "for research use only" to bypass these laws. While enforcement usually targets sellers, individuals who purchase these products have no guarantee of their quality, purity, or safety and have zero consumer protection (PeptideClinicLocator.com). The only legal and safe route is through a medical provider and a licensed pharmacy.
Why is TB-500 still restricted by the FDA?
TB-500 (Thymosin Beta-4) is expected to remain on the Category 2 list, meaning it will likely stay prohibited from compounding. According to industry reports, this is due to unaddressed safety concerns, particularly around the potential for cell proliferation (PeptideMark). Without more robust human safety data to mitigate these concerns, the FDA is unlikely to change its status, distinguishing it from other peptides like BPC-157.
Contributing to evidence-based peptide education and provider transparency.
Too many clinics, not enough clarity?
We don't sell peptides. Tell us your goals and we'll connect you with a vetted provider who can figure out what actually makes sense for you.
See what's right for youRelated articles
How Protein and Muscle Growth Actually Work
It's not just about eating more. Understand the real science of protein and muscle growth to learn how much you need and the best timing to see results.
Peptide ScienceWhat are the best peptides for muscle growth and fat loss?
Exploring peptides for muscle growth and fat loss? Discover the most effective options for body recomposition, how they actually work, and what to expect.
Peptide ScienceThe Key L-Lysine Benefits for Skin, Immunity & Stress
Discover the science-backed l-lysine benefits. Our guide covers how this essential amino acid may boost collagen, fight viruses, and aid calcium absorption.