Are the Ozempic Side Effects Really That Bad?
From nausea to more serious risks, we cover the full spectrum of Ozempic side effects. Learn what to expect, when to worry, and how to manage them.

This content is for informational purposes only and does not constitute medical advice. Always consult with a qualified healthcare provider for any health concerns or before starting a new treatment.
GLP-1 medications like Ozempic have become incredibly popular for managing type 2 diabetes and, off-label, for weight loss. But with their rise in use, it's crucial to have a clear understanding of the potential Ozempic side effects. As of 2026, the information available has grown significantly, thanks to ongoing research and updates from regulatory bodies like the FDA. We'll break down what you need to know, from the most common issues to the more serious warnings, and touch on the side effects of Mounjaro and Zepbound for comparison.
Common Ozempic Side Effects
Most people who experience side effects from Ozempic find they are gastrointestinal (GI) in nature. These issues are most common when you first start the medication or after a dose increase. This happens because Ozempic's active ingredient, semaglutide, works in part by slowing down how quickly your stomach empties.
The good news is that these side effects often decrease over time as your body adjusts. This is why healthcare providers recommend a gradual dose escalation, typically starting at 0.25 mg weekly and slowly increasing to a maintenance dose of 0.5 mg, 1 mg, or 2 mg (FDA, January 2025).
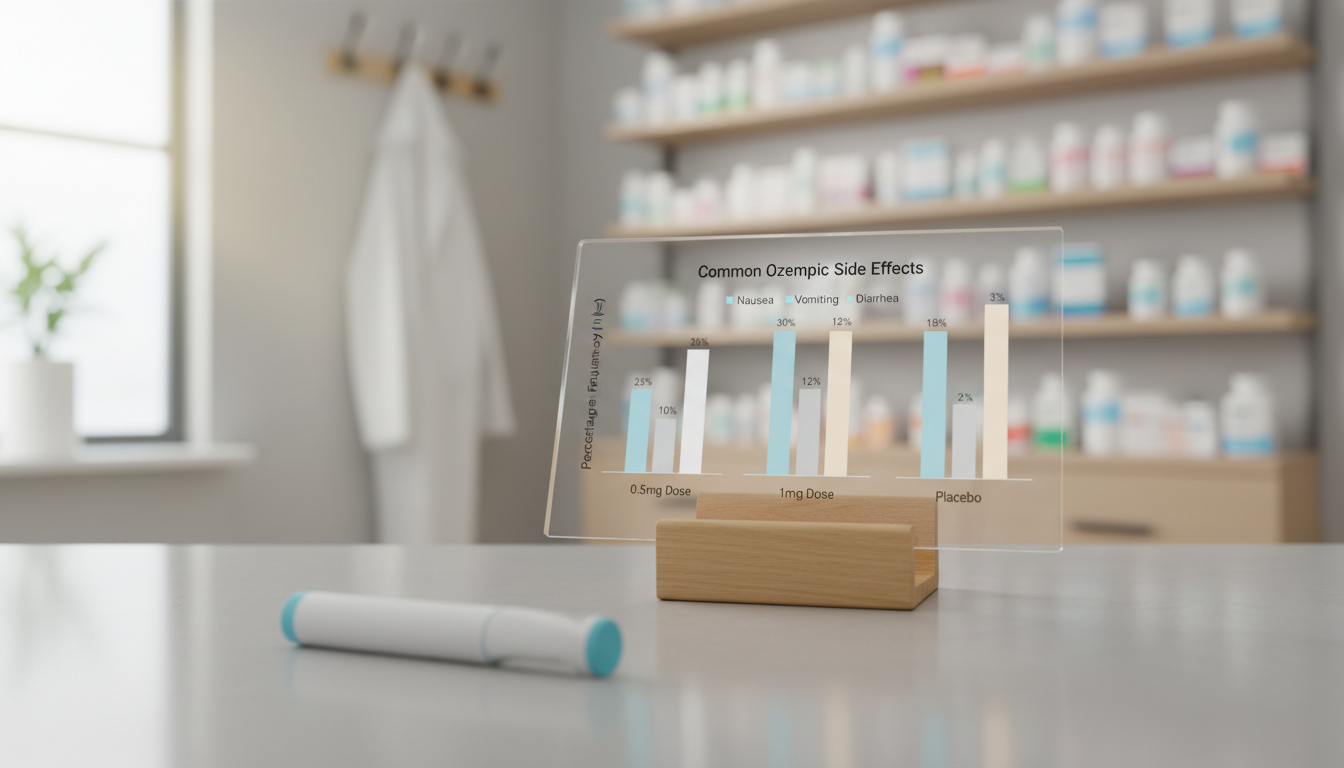
Here’s a look at the frequency of common Ozempic side effects reported in clinical trials compared to a placebo, based on the official prescribing information.
| Adverse Reaction | Ozempic 0.5 mg | Ozempic 1 mg | Placebo |
|---|---|---|---|
| Nausea | 16% | 20% | 6% |
| Vomiting | 5% | 9% | 2% |
| Diarrhea | 8% | 8% | 2% |
| Abdominal Pain | 7% | 6% | 4% |
| Constipation | 3% | 5% | 1% |
Source: HIGHLIGHTS OF PRESCRIBING INFORMATION for Ozempic, October 2025.
As you can see, nausea is the most frequently reported issue. It affects up to one in five people on the 1 mg dose. Usually, these GI symptoms are mild to moderate. With the recent FDA approval of an oral Ozempic pill, expected in the second quarter of 2026, patients will have another option, but similar GI side effects are anticipated (Drugs.com, February 2026).
Learning about these potential effects can be a lot to process. If you're trying to figure out if a GLP-1 medication is the right fit for your health goals, expert guidance is key.
Not sure which treatment is right for you? Take our free 3-minute quiz to get matched with a licensed provider who can help weigh the pros and cons for your specific situation.
Serious Ozempic Side Effects to Watch For
While most side effects are manageable, the FDA has updated Ozempic's label through 2025 to include several serious warnings. It's important to be aware of these so you can seek medical attention if necessary.
Thyroid C-Cell Tumors
This is the most prominent warning and comes with a "boxed warning," the FDA's strongest type. In rodent studies, semaglutide caused thyroid C-cell tumors. It is still unknown if Ozempic causes these tumors, including a type of thyroid cancer called medullary thyroid carcinoma (MTC), in humans (FDA, January 2025). Because of this risk, Ozempic should not be used by people with a personal or family history of MTC or by those with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Severe Gastrointestinal Reactions
Beyond common nausea, the FDA has expanded warnings to include more severe GI issues. These include ileus, which is a lack of movement in the intestines that can lead to a buildup and blockage of food. The label updates warn about intestinal obstruction and gastroparesis, also known as stomach paralysis (DefectiveDrugSite.com, December 2025). If you have a history of severe gastroparesis, Ozempic is not recommended.
Acute Pancreatitis
Inflammation of the pancreas, or pancreatitis, has been reported in patients taking Ozempic. In clinical trials, confirmed cases occurred in 7 Ozempic-treated patients versus 3 in patients on other therapies (FDA, 2023). Stop using Ozempic and call your doctor immediately if you experience severe pain in your stomach area that won’t go away, with or without vomiting. You may feel the pain radiate from your abdomen to your back.
Diabetic Retinopathy Complications
In a two-year trial of patients with type 2 diabetes and high cardiovascular risk, more patients treated with Ozempic reported complications related to diabetic retinopathy (3.0%) compared to placebo (1.8%) (FDA, 2023). The risk is higher in patients with a history of diabetic retinopathy. Rapid improvement in blood sugar control can sometimes temporarily worsen this condition.
Kidney Injury
Ozempic can cause or worsen kidney problems. Acute kidney injury has been reported, sometimes requiring dialysis, often in patients who experienced severe nausea, vomiting, or diarrhea that led to dehydration (DefectiveDrugSite.com, December 2025). Staying well-hydrated is critical.
Gallbladder Problems
Acute gallbladder disease, including gallstones and inflammation of the gallbladder (cholecystitis), has occurred in patients taking Ozempic. Some cases have required hospitalization and gallbladder removal.
Risk of Aspiration During Surgery
A newer warning added in late 2024 and highlighted in 2025 advises patients about the risk of pulmonary aspiration during general anesthesia or deep sedation. Because Ozempic delays stomach emptying, there's a risk that stomach contents could be inhaled into the lungs. You must inform your healthcare team that you are taking Ozempic before any planned surgery or procedure (FDA, November 2024).
It’s worth noting that in March 2026, the FDA issued a warning letter to Novo Nordisk, the maker of Ozempic, for allegedly failing to report some serious adverse events in a timely manner. While the letter noted the events were not necessarily caused by the drug, it underscores the importance of transparent reporting (kcra.com).
Who Should Not Take Ozempic or Similar GLP-1s?
GLP-1 medications are not suitable for everyone. You should not use Ozempic, Mounjaro, or Zepbound if you have certain medical conditions. Always discuss your full health history with your provider.
Key contraindications include:
- Personal or family history of Medullary Thyroid Carcinoma (MTC): Due to the boxed warning for thyroid C-cell tumors, this is a firm contraindication.
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2): This genetic condition also increases the risk of MTC.
- A previous serious allergic reaction to semaglutide or tirzepatide: If you've had a hypersensitivity reaction, like anaphylaxis or angioedema, to the active ingredient or any other component in the injection, you should not take it.
- History of severe gastroparesis: Since these drugs slow stomach emptying, they can worsen this condition significantly (FDA, January 2025).
Additionally, caution is advised for patients with a history of:
- Pancreatitis: While not an absolute contraindication, a prior history of pancreatitis may increase your risk.
- Diabetic Retinopathy: Patients with a history of this eye condition should be monitored closely.
- Severe Kidney Disease: Your provider will need to carefully consider the risks and benefits. Ozempic is indicated to reduce the risk of kidney disease progression in some diabetic patients, but careful monitoring is still needed (FDA, October 2025).
These medications have also not been studied in pregnant or breastfeeding women, so they are not recommended in those situations.
Drug Interactions with Ozempic
When you start a new medication, it's important to consider how it might interact with other drugs you're taking. The main interaction concern with Ozempic and other GLP-1s stems from their primary mechanism of action.
Because Ozempic slows down gastric emptying, it can affect the absorption of oral medications taken at the same time. This could make other pills less effective because they move through your system differently. Your provider may advise you to time your oral medications carefully in relation to your Ozempic injection or monitor their effects more closely.
Another significant interaction is with other medications used to manage blood sugar, such as:
- Insulin: Taking Ozempic with insulin can increase your risk of hypoglycemia (low blood sugar). Your provider will likely need to adjust your insulin dose when you start Ozempic.
- Sulfonylureas (e.g., glipizide, glyburide): These drugs also lower blood sugar, and combining them with Ozempic significantly raises the risk of hypoglycemia. A dose reduction of the sulfonylurea is often necessary.
Always give your doctor and pharmacist a complete list of all medications you take, including prescription drugs, over-the-counter medicines, vitamins, and herbal supplements. You can also use online tools as a preliminary reference, such as RxFox's drug interaction checker, before discussing with your provider.

How to Manage Common Side Effects
Dealing with GI side effects can be discouraging, but there are practical steps you can take to minimize them, especially during the initial weeks of treatment.
For Nausea:
- Eat smaller, more frequent meals. A large, heavy meal is more likely to sit in your stomach and cause nausea.
- Choose bland, low-fat foods. Think toast, crackers, rice, and baked chicken. Avoid greasy, fried, or very sweet foods. Our guide on foods to avoid on semaglutide has more tips.
- Eat slowly and stop when you feel full. Ozempic helps you feel full faster. Listen to those signals.
- Stay hydrated. Sip water or clear, cold drinks throughout the day.
- Avoid lying down right after eating.
For Diarrhea:
- Focus on hydration. Diarrhea can lead to dehydration. Drink plenty of fluids, and consider drinks with electrolytes if it's severe.
- Incorporate soluble fiber. Foods like bananas, oatmeal, and applesauce can help firm up stools.
- Temporarily avoid high-fiber trigger foods like raw vegetables or beans until the issue resolves.
For Constipation:
- Increase your fluid intake. Water is essential for helping fiber work and keeping stools soft. Read more about managing Ozempic constipation.
- Gradually increase your fiber intake. Focus on sources like whole grains, fruits with skin, and vegetables.
- Stay active. Physical activity can help stimulate bowel movements. A daily walk can make a big difference.
For Abdominal Pain: Mild abdominal cramping can occur. Use the tips for nausea and try to relax. However, if the pain is severe, persistent, or radiates to your back, it could be a sign of pancreatitis, and you should contact your doctor immediately.
Managing these initial hurdles can be the key to long-term success with treatment. If you're struggling, don't hesitate to seek professional support.
Want to see which options fit your budget and goals? Our free quiz takes 3 minutes and matches you with vetted providers who can create a management plan for you.
When to See a Doctor
While mild side effects can often be managed at home, certain symptoms require immediate medical attention. Contact your healthcare provider or seek emergency care if you experience any of the following:
- Severe abdominal pain that is persistent, sharp, and may radiate to your back (potential sign of pancreatitis).
- Signs of a gallbladder problem, such as pain in your upper right stomach, fever, jaundice (yellowing of the skin or eyes), or clay-colored stools.
- Symptoms of a serious allergic reaction, including swelling of your face, lips, tongue, or throat; problems breathing or swallowing; severe rash or itching; fainting or feeling dizzy.
- Vision changes, especially if you have a history of diabetic retinopathy.
- A lump or swelling in your neck, hoarseness, trouble swallowing, or shortness of breath (potential signs of a thyroid tumor).
- Persistent vomiting or diarrhea that prevents you from keeping liquids down, which could lead to severe dehydration and kidney problems.
Always err on the side of caution. Your doctor is your best resource for determining if a symptom is a normal side effect or a sign of a more serious issue.
FAQs
Does Mounjaro have less side effects than Ozempic?
Both medications have very similar gastrointestinal side effect profiles, including nausea, vomiting, and diarrhea. Some clinical data suggests that tirzepatide (the active drug in Mounjaro and Zepbound) may have slightly higher rates of these GI issues at comparable doses. A large meta-analysis confirmed that tirzepatide was associated with an increased risk of these common GI events (BMC Medicine, 2026). However, individual experiences vary greatly, and what matters most is how your body responds. The key question is not just about the side effects but finding the best online GLP-1 provider to guide you.
What are the Mounjaro long term side effects?
Since tirzepatide is newer than semaglutide, data on Mounjaro long term side effects is still emerging. However, like Ozempic, Mounjaro and Zepbound carry a boxed warning for the risk of thyroid C-cell tumors based on rodent studies. The other known serious potential side effects, like pancreatitis, gallbladder disease, and kidney injury, are similar to those seen with other GLP-1 receptor agonists. Ongoing studies will provide more clarity on tirzepatide long-term side effects over time.
What side effects can I expect when switching from Ozempic to Mounjaro?
When switching from Ozempic to Mounjaro, your provider will start you on a low dose of tirzepatide and titrate up, just as you did with Ozempic. You may experience a temporary return of GI side effects like nausea or diarrhea as your body adjusts to the new medication. This is a common part of the process when changing between GLP-1 medications.
Are there any positive or unexpected side effects?
Interestingly, yes. A massive 2026 study found that GLP-1 medications like semaglutide may have surprising benefits for mental health. The research showed that users had a significantly lower risk of developing depression (44% lower) and anxiety (38% lower). It also noted a 47% reduction in hospitalizations related to substance use disorders (Newsweek, 2026). While the mechanism isn't fully understood, it's a promising area of ongoing research.
What are the main side effects of Ozempic and Mounjaro?
The primary side effects of Ozempic and Mounjaro are gastrointestinal. The most common are nausea, diarrhea, vomiting, decreased appetite, constipation, and abdominal pain. These are most prevalent when starting the medication or increasing the dose and typically lessen over time for most people.
Ready to explore your options with a professional? Take our free 3-minute quiz to get matched with a licensed provider who can help you find the right path forward.
Contributing to evidence-based peptide education and provider transparency.
Too many clinics, not enough clarity?
We don't sell peptides. Tell us your goals and we'll connect you with a vetted provider who can figure out what actually makes sense for you.
See what's right for youRelated articles
A Step-by-Step Gelatin Recipe for Weight Loss
Seen the viral "pink trick"? We break down the simple gelatin recipe for weight loss. Learn our step-by-step method to help manage hunger and feel full.
GLP-1 Weight LossOzempic Cost in 2026: What You'll Actually Pay
The 2026 Ozempic cost can be anywhere from $25 to over $1,400 a month. Discover why and learn how to find savings with or without insurance coverage.
GLP-1 Weight LossHow Zep Weight Loss Works and What to Expect
Thinking about zep weight loss? We cover how this drug works, what realistic results look like, common side effects, and how it compares to Ozempic.